|
The occurrence and natural abundances of isotopes can be experimentally determined using an instrument called a mass spectrometer. And the methods of using isotopes for this research contributed to entire fields of study. One of her first discoveries provided insight into the phosphorylation of glucose that takes place in mitochondria. Mildred Cohn pioneered the usage of these isotopes to act as tracers, so that researchers could follow their path through reactions and gain a better understanding of what is happening. Oxygen-18 was also critical to the discovery of metabolic pathways and the mechanisms of enzymes. It is important in paleoclimatology, for example, because scientists can use the ratio between Oxygen-18 and Oxygen-16 in an ice core to determine the temperature of precipitation over time. Let's consider just one natural, stable isotope: Oxygen-18, which is noted in the table above and is referred to as one of the environmental isotopes. The elements in group 1 are known as the alkali metals those in group 2 are the alkaline earth metals those in 15 are the pnictogens those in 16 are the chalcogens those in 17 are the halogens and those in 18 are the noble gases.Īs you will learn, isotopes are important in nature and especially in human understanding of science and medicine. Groups are numbered 1–18 from left to right. Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals. Elements in the same group of the periodic table have similar chemical properties. The discovery of the periodic recurrence of similar properties among the elements led to the formulation of the periodic table, in which the elements are arranged in order of increasing atomic number in rows known as periods and columns known as groups. A chemical symbol identifies the atoms in a substance using symbols, which are one-, two-, or three-letter abbreviations for the atoms. When a naturally occurring element is composed of several isotopes, the atomic mass of the element represents the average of the masses of the isotopes involved. Isotopes of an element are atoms with the same atomic number but different mass numbers isotopes of an element, therefore, differ from each other only in the number of neutrons within the nucleus. View available translations of the content. Download the content in PDF, Microsoft Word, or other format. Questions and Tasks Add a note to the content. Kinetic Molecular Theory, Real GasesĬontent Foreword Atoms Introduction Matter Measurement The Atom Periodic Table Moles & Mass Light Blackbody Radiation, Photoelectric Effect Atomic Spectra, Bohr Model Orbitals & Quantum Numbers Electron Configurations Periodic Trends Molecules Bonding Nomenclature Lewis Structures Part 1 Lewis Structures Part 2 Molecular Shape Polarity Organic Molecules Isomers Valence Bond Theory Molecular Orbital Theory Interactions Pressure & Gas Laws Combined & Ideal Gas Laws Dalton's Law, Graham's Law, Henry's Law Kinetic Molecular Theory, Real Gases Intermolecular Forces Properties of Water Applications of IMF Phase Diagrams Reactions Stoichiometry Limiting Reactants, % Yield % Composition, Empirical Formulas Energy, Heat, and Work Calorimetry Part 1 Calorimetry Part 2 Enthalpy Part 1 Enthalpy Part 2 Entropy Gibb's Free Energy Climate and Human Impacts Search within this publication Search the entire site 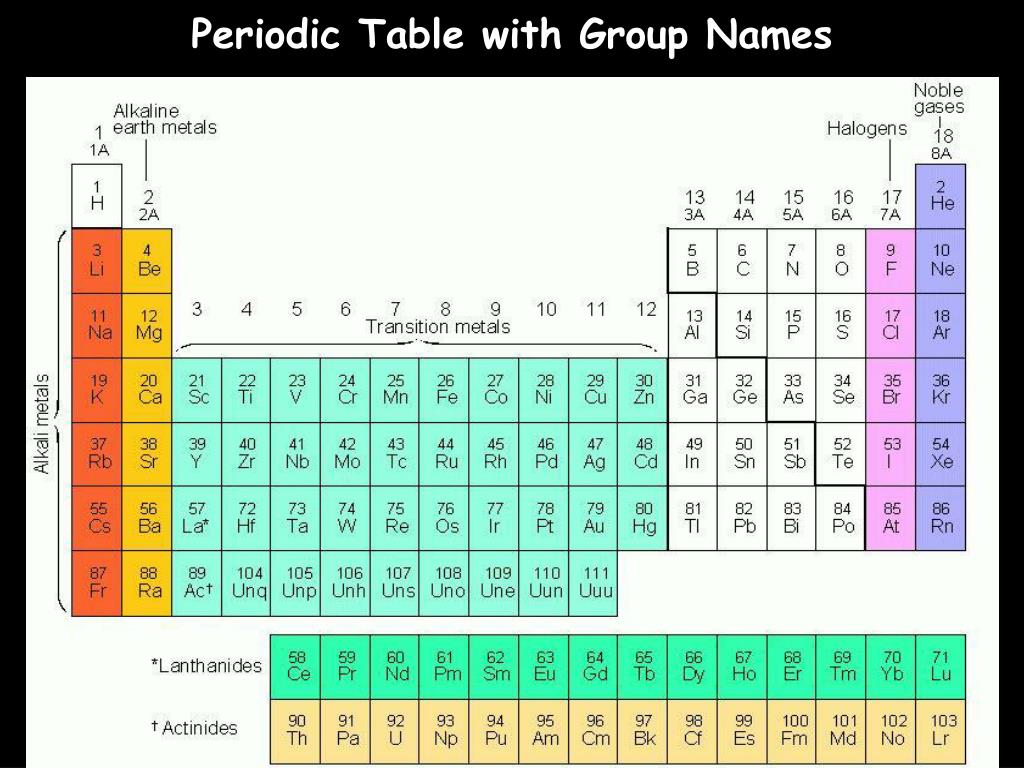
Blackbody Radiation, Photoelectric Effect
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
